 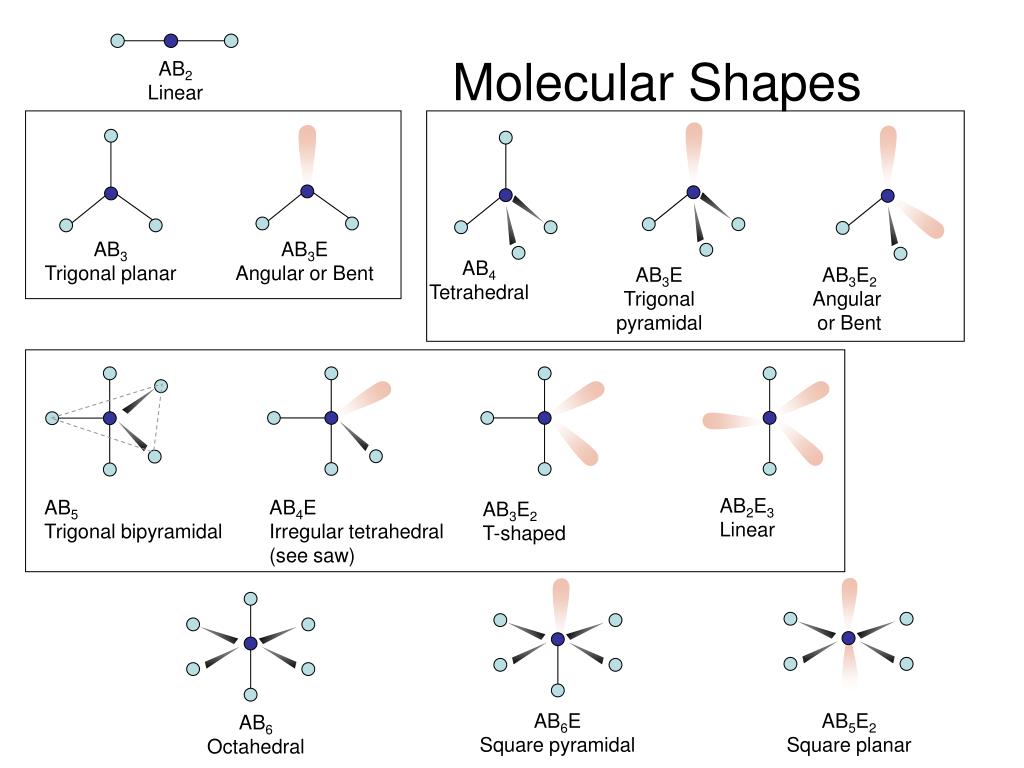
In short, the repulsion between the pairs of valence electrons of all the atoms in the molecule determines the geometry of the resulting molecule.The aim is that the molecule must have minimum energy and maximum stability.The Valence Shell Electron Pair Repulsion Theory (VSEPR) states that “Whenever there is a repulsion between the pairs of valence electrons in all atoms, the atoms will arrange themselves in a geometric shape so as to minimize the electron pair repulsion.”.The shape of the atom is dependent upon the repulsion between the pairs of valence electrons. The attraction force between nucleus and electrons and the repulsion force between the electrons. At this point, two types of forces come into play. Atoms bond chemically in order to form a molecule.It could not give any information about the angles or lengths of bonds in 3D.Ĭheck more important topics of Chemistry here.It could not give any information about the nature of attractive forces between the constituent atoms of a molecule.It could not give the shapes of molecules.It could not give any information about the release of energy during the structural formation of a covalent bond.In the case of Resonance structure, it isn’t possible to produce two distinct Lewis electronic structures for the same atomic composition.According to the Lewis Structure, the same atomic composition can correspond to many acceptable Lewis structures.This theory was used to predict the shapes of many molecules and polyatomic ions.It was called the Valence Shell Electron Pair Repulsion (VSEPR) model.However, it wasn’t enough to give a complete understanding of the molecules’ shape.It gave the structure of the molecule and information about which atoms have lone pairs of electrons.At the start, the Lewis electron-pair structure was used to predict the number and types of bonds between the atoms of a molecule.Failure of Lewis Structure and Need of VSEPR Theory Read more about Blocks of Periodic Table, here.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
